Organic certification rarely fails because teams misunderstand the rules. It fails because systems are not aligned across markets.
For Quality Managers overseeing EU Organic, USDA NOP, JAS, or other regional schemes, the complexity is not regulatory literacy. It is structural coherence.
Based on aggregated FoodChain ID certification experience across multiple regions, multi-market organic findings most often stem from fragmented documentation logic, inconsistent scope governance, and uncoordinated audit planning rather than lack of expertise.
Organic compliance is no longer local
Operations may be site-specific, but certification oversight is increasingly global. A product approved in one jurisdiction may require documentation adjustments, labeling interpretation, or scope updates in another.
Over time, regional differences accumulate. Scope definitions evolve independently. Supplier documentation is stored by market instead of by control logic. Audit calendars overlap. Label governance becomes decentralized.
What begins as manageable variation gradually reduces predictability.
From a system-level perspective, fragmentation weakens inspection confidence before it weakens compliance.
Where alignment typically breaks down
Across anonymized certification observations, alignment gaps most commonly appear in three structural areas.
1. Scope governance and operational drift
As suppliers, storage locations, or trading activities evolve, scope documentation is not always updated consistently across all organic schemes. Inspections then focus on reconciling declared scope with operational reality, creating avoidable corrective actions.
The issue is rarely technical non-compliance. It is asynchronous system updates across jurisdictions.
2. Documentation sequencing and transaction certificate flow
One structural insight frequently observed in multi-market organic programs involves transaction certificate management.
In some regions, transaction certificates are required more systematically. In others, reliance is placed primarily on annual certification and mass balance reconciliation. When these documentation flows are not harmonized, cross-market inconsistencies emerge.
For example, shipment documentation may be compliant locally but misaligned with centralized mass balance records. During inspection, auditors then spend time validating reconciliation logic rather than reviewing control effectiveness.
This is not a documentation volume problem. It is a sequencing problem.
3. Mass balance pressure under growth
The 2025 organic roadmap work highlighted a recurring structural tension: mass balance logic often remains market-specific while sourcing becomes global.
As organizations expand supplier networks across regions, raw material inflows may be recorded differently per market. Without a centralized reconciliation methodology, periodic mass balance reviews become reactive instead of predictive.
From an inspection standpoint, mass balance discrepancies rarely stem from fraud. They more often reflect inconsistent recording logic across markets.
When organic oversight scales without structural alignment, reconciliation risk increases.
None of these issues indicate weak controls. They indicate disconnected system architecture.
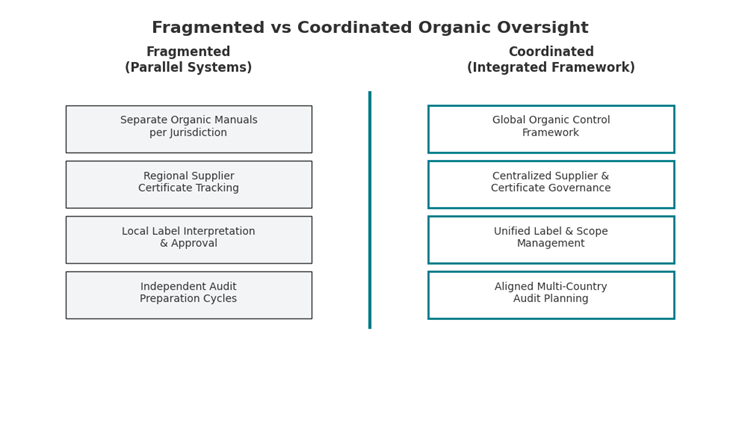
Before and After: Structural Alignment in Practice
Across recurring certification patterns, the difference between fragmented and coordinated organic oversight is structural rather than regulatory.
Before: Parallel National Systems
Organic compliance is managed per jurisdiction. Documentation repositories are regional. Supplier certificate tracking and transaction documentation follow local practices. Mass balance reconciliation occurs independently per market. Audit preparation cycles are not aligned.
In this structure, inspections often focus on reconciling documentation flows instead of validating preventive controls. Repeat findings tend to cluster around scope clarity, certificate consistency, and reconciliation timing.
After: Coordinated Multi-Country Framework
A single global organic control framework governs scope, supplier evidence, transaction documentation flow, and mass balance methodology. Local regulatory differences are layered onto one structured system. Documentation is centralized and version-managed. Corrective actions follow unified governance logic.
In this model, inspection discussions shift from document reconciliation toward control effectiveness. Scope clarifications decrease. Preparation cycles shorten because evidence is structured coherently across markets. Documentation-related repeat findings reduce. These observations reflect aggregated certification experience rather than individual client data.

Why this matters more in 2026
Regulators and buyers increasingly assess organic certification as a control system rather than a checklist. Traceability depth, mass balance defensibility, and documentation governance receive greater scrutiny across markets.
Fragmented oversight may function in early growth stages. It rarely scales cleanly across jurisdictions.
For Global Quality and Compliance Leaders, the question is no longer whether each region holds certification. It is whether the organic framework is globally coherent and defensible under inspection pressure.
Organic compliance is strongest when it operates as one structured process across every market.
Explore how coordinated multi-country Organic Certification simplifies oversight.


