For Quality Managers in the animal feed sector, certification audits rarely fail because of missing intent or effort.
They fail because systems don’t scale, controls aren’t connected, or planning happens too late.
Across EU and national feed certification audits — including GMP+, FCA, OQUALIM, CSA-GTP, and related schemes — the same operational pitfalls appear repeatedly, even in mature organisations. The difference between plants that struggle and those that perform consistently is not “more compliance”, but better system design and predictability. Below are the five most common feed certification pitfalls we see in audits, illustrated with plant-level benchmarks, and the practical actions Quality Managers take to prevent them.
Pitfall #1: Managing certifications as separate programs instead of one system
What we see in audits
Many feed operations manage GMP+, national schemes, non-GMO modules, and sustainability requirements as parallel tracks. Each standard has its own procedures, records, and corrective actions.
This results in:
- duplicated documentation
- inconsistent terminology
- longer audits
- repeat non-conformances linked to system coherence
Why this becomes a real risk
Auditors do not audit standards — they audit systems. When requirements are fragmented, auditors spend more time reconciling evidence than validating controls. This issue becomes particularly visible in multi-site or multi-country operations, where inconsistencies between sites lead to uneven audit outcomes and repeated findings across the group.
What high-performing plants do differently
They design one feed safety and compliance system, with certification requirements layered on top.
Benchmark we consistently observe:
Plants that move from siloed certification management to a unified system typically achieve:
- 20–30% fewer on-site audit days across combined schemes
- 25%+ reduction in repeat non-conformances linked to documentation and system consistency
- Faster audit close-out because corrective actions apply across standards
A simple system map that works
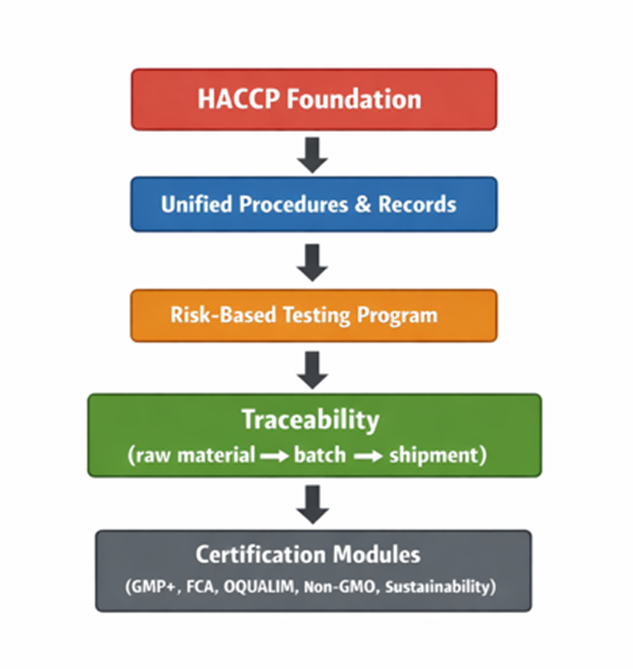
When auditors can follow this logic end-to-end, audits become shorter, clearer, and more predictable.
Pitfall #2: Incomplete or outdated certification scope
What we see in audits
Scope statements often fail to reflect:
- trading vs manufacturing activities
- transport and storage responsibilities
- outsourced or newly added operations
Even well-controlled plants receive findings because the declared scope no longer matches operational reality.
Why this matters
Feed certifications are role-specific. A mismatch between real activities and declared scope leads to:
- scope corrections during audits
- follow-up visits
- avoidable non-conformances
How Quality Managers prevent this
High-performing teams:
- formally review scope whenever operations change
- align scope definitions across all certifications
- validate scope during internal audits, not at the certification audit
This simple discipline reduces scope-related findings, one of the most common — and preventable — audit issues.
At group level, late audit planning often creates bottlenecks across multiple sites simultaneously, amplifying resource strain and reducing global audit predictability. This applies to leaders overseeing portfolios, not just plants.
Pitfall #3: Weak traceability for high-risk raw materials
What we see in audits
Traceability systems often function well for standard ingredients but break down for high-risk materials, such as:
- soy and soy derivatives
- imported proteins
- materials linked to non-GMO or sustainability claims
Common gaps include incomplete supplier documentation or batch records that cannot be demonstrated quickly during audits.
Why this is increasingly critical
EU feed regulations, RASFF alerts, EUDR expectations, and sustainability verification all require traceability depth, not just traceability existence .
A practical audit-ready traceability check
Quality Managers in high-performing plants can answer “yes” to all of the following for high-risk materials:
- Batch-level traceability from supplier to finished feed
- Supplier documentation aligned with current certification scope
- Testing results linked to specific batches (not generic COAs)
- Clear escalation process for missing or inconsistent data
- Traceability verified during internal audits, not only external ones
Plants that formalize this checklist typically experience fewer major findings and faster auditor verification.
Pitfall #4: Treating testing as a reactive requirement
What we see in audits
Testing programs are often designed to:
- meet minimum certification requirements
- respond to incidents or complaints
This reactive approach disconnects testing from preventive risk control.
Why this leads to repeat findings
Auditors increasingly expect testing to be:
- risk-based
- linked to HACCP
- adapted to seasonality and supplier performance
When testing is not integrated into preventive controls, the same findings recur.
What effective plants do
They integrate testing into risk management by:
- adjusting frequency based on raw material risk
- aligning testing plans with HACCP reviews
- using results to drive corrective and preventive actions
This approach consistently reduces repeat non-conformances related to contaminants and supplier control.
Pitfall #5: Late audit planning and resource bottlenecks
What we see in audits
Audits scheduled late in the year or rescheduled multiple times create:
- rushed preparation
- limited auditor availability
- longer on-site audit days
Why this increases audit effort
Late planning compresses preparation time and reduces flexibility for both auditors and QA teams.
Benchmark from audit planning practice
Quality Managers who schedule audits 6–9 months in advance typically experience:
- Fewer cancellations and reschedules
- Shorter on-site audit duration
- More predictable audit outcomes
- Reduced internal workload during peak production periods
Early planning functions as a risk-reduction control, not an administrative task.
A practical tool to prevent these pitfalls before your next audit
Most of the issues described above don’t require more documentation — they require better alignment between scope, systems, testing, and planning.
To help Quality Managers translate these lessons into action, we’ve created a Feed Certification Audit-Readiness Checklist that can be used as a pre-audit scope and system review.
The checklist helps you verify, in one structured review:
- whether your certification scope reflects actual operations
- if multiple certifications are managed as one coherent system
- whether high-risk raw materials have sufficient traceability depth
- if testing supports preventive control
- whether audits are planned early enough to avoid bottlenecks
Plants that apply this review 90–120 days before audits typically experience fewer audit days, fewer repeat findings, and more predictable outcomes.
Download the Feed Certification Audit-Readiness Checklist
A practical scope and system review tool Quality Managers use to prepare audits before issues surface.
What these pitfalls have in common
None of these issues stem from lack of expertise or commitment.
They stem from fragmentation, late decisions, and disconnected controls.
High-performing feed operations treat certification as:
- a system, not a checklist
- a preventive tool, not a reactive obligation
- a strategic enabler of market access and trust
What this means for Global Quality & Compliance Leaders
For Quality leaders responsible for multiple sites, countries, or certification schemes, these pitfalls rarely appear in isolation. They compound across the organization.
In global or multi-site operations, fragmented certification management often leads to:
- inconsistent audit outcomes between sites
- uneven interpretation of requirements
- duplicated audit effort
- repeated non-conformances that migrate from one site to another
Global Quality & Compliance Leaders who standardize certification structure, scope logic, and audit planning principles across sites typically achieve greater consistency in audit outcomes and better predictability at group level, even when local schemes differ.
The challenge is not enforcing identical documentation everywhere — it is ensuring one coherent system logic that scales across sites.
Final takeaway for Quality Managers
Avoiding audit findings does not require more paperwork.
It requires better alignment between:
- certification structure
- testing strategy
- traceability depth
- audit planning discipline
Plants that take this approach consistently:
- reduce audit days
- limit repeat non-conformances
- regain predictability across certification cycles
Want to benchmark your certification approach?
See how feed Quality Managers reduce audit effort and non-conformances.
Benchmark how integrated feed certification programs help reduce audit days by up to 20–30% and cut repeat non-conformances by 25%+ through better planning and scheme alignment.
Discuss your certification benchmark with an FCID specialist



