For Quality Managers in the feed sector, audits rarely become difficult because of technical gaps. They become difficult because planning happens too late.
Late audit scheduling creates a chain reaction:
- limited auditor availability
- rushed preparation
- compressed corrective action windows
- longer on-site audit days
- repeat non-conformances that could have been avoided
Across feed manufacturing, trading, and storage operations, one pattern consistently separates high-performing sites from stressed ones:
Quality Managers who plan feed audits 9+ months in advance achieve more predictable audits, fewer repeat findings, and lower operational disruption.
This article explains how to plan feed audits proactively, what to align internally, and why early planning is one of the most effective risk-reduction controls available to Quality Managers.
Why late audit planning is a hidden risk
In many organisations, audit scheduling is treated as an administrative task — something triggered when certificates are about to expire or customer requests arrive.
In reality, audit planning directly affects:
- system readiness
- documentation quality
- staff availability
- auditor allocation
- audit scope accuracy
When planning is late, even strong systems can appear weak under audit pressure.
Observed benchmark across feed operations we assess regularly:
Plants that schedule audits early typically experience shorter on-site audits (20–30% fewer audit days) and lower rates of repeat non-conformances, simply because preparation is controlled instead of reactive.
Step 1: Lock the certification strategy before locking dates
Audit planning should start with strategy, not calendars.
Before requesting audit dates, confirm:
- which certifications are required (GMP+, FCA, OQUALIM, CSA-GTP, non-GMO, sustainability modules)
- whether certifications can be combined or sequenced
- which sites and activities fall under each scope
Common pitfall: scheduling separate audits for certifications that could have been integrated, increasing audit days and internal workload.
What effective Quality Managers do:
They define a certification roadmap for the year, then plan audits to support that roadmap — not the other way around.
Step 2: Align scope early to avoid last-minute corrections
One of the most frequent causes of audit delays is scope misalignment discovered too late.
Early in the planning cycle, Quality Managers should validate:
- whether trading, storage, transport, or outsourced activities are in scope
- whether new products, suppliers, or processes have been added
- whether scope descriptions are consistent across all certifications
When scope is aligned months in advance, audits:
- start on time
- require fewer clarifications
- avoid corrective visits linked to scope errors
Step 3: Map internal readiness milestones backwards from the audit date
High-performing teams don’t ask “Are we ready?” one month before the audit.
They work backwards from the audit date.
A practical planning sequence looks like this:
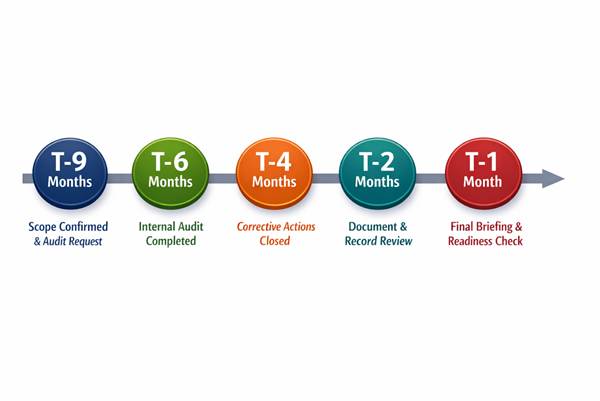
This cadence transforms audits from a disruptive event into a controlled verification.
| “This is usually the point where audit planning becomes unclear. To avoid missing scope, timing, or documentation gaps, many Quality Managers use a structured audit-readiness checklist before audit dates are locked.” |
Step 4: Secure auditor availability early (especially for peak periods)
Feed audits often cluster around:
- harvest seasons
- year-end customer deadlines
- combined certification windows
When audits are requested late, auditor choice is limited — sometimes forcing:
- split audits
- extended audit duration
- suboptimal scheduling
Planning 9+ months ahead allows:
- access to the most experienced auditors
- smoother multi-site coordination
- better alignment with production schedules
This directly contributes to shorter audits and fewer findings, because auditors have the time to assess systems properly.
Step 5: Integrate audit planning with testing and supplier risk cycles
Audit planning should not exist in isolation.
Quality Managers who plan effectively:
- align audit timing with raw material risk periods
- ensure testing data is available and reviewed before audits
- avoid audits during known operational stress points
When testing, supplier performance, and audits are aligned, audits become:
- more focused
- easier to demonstrate
- less corrective-action heavy
What early audit planning changes in practice
Across feed operations that adopt early audit planning as a standard practice, we consistently see:
- 20–30% reduction in on-site audit days
- fewer repeat non-conformances, particularly around documentation and traceability
- more predictable audit outcomes, reducing stress on QA teams
- lower disruption to production and logistics
These improvements are not driven by additional resources — they are driven by timing and coordination.
Final takeaway for Quality Managers
Audit planning is not a scheduling task. It is a risk-management decision.
By planning feed audits 9+ months ahead, Quality Managers:
- regain control over timing and scope
- reduce audit effort
- improve consistency across certification cycles
- protect their teams from last-minute firefighting
In an environment of increasing regulatory scrutiny, predictability is a competitive advantage.
A practical next step
Before confirming your next audit window, validate readiness across scope, systems, testing, and planning.
To support early planning, we’ve created a Feed Certification Audit-Readiness Checklist that Quality Managers use to assess readiness before audit dates are locked.


